ASIAN PATENT ATTORNEYS ASSOCIATION
|
|
|
- May Maxwell
- 6 years ago
- Views:
Transcription
1 ASIAN PATENT ATTORNEYS ASSOCIATION 63 rd Council Meeting Penang, Malaysia Patent Committee Report: INDIA Hari Subramaniam & Calab Gabriel 1
2 India: Patents 2014 There have been several changes in statutory law and in regulations since we last presented our report in Hanoi in The past one year has again seen several judicial pronouncements by various High Courts and by the Intellectual Property Appellate Board (IPAB) that have resulted in significant changes in practice due to interpretation and clarification of existing statutory provisions. The IPAB and the High Courts of New Delhi, Mumbai and Chennai have been active in patent law interpretation on both procedural and substantive issues. These decisions have resulted in certainty in some areas of practice and caused a bit of concern in respect of the others. Some of these decisions are discussed below: I Legislative Update 1) Patent Rules amended Effective February 28, 2014, the new Patents (Amendment) Rules 2014 have come into force. The new Rules primarily consist of revision in the existing fee structure across all actions. A brief summary of the Rules is as follows: A new category of applicant, namely, small entity and a new Form applicable thereto have been introduced. For the purposes of the amendment, small entity shall be interpreted as per the Micro, Small and Medium Enterprises (MSME) Development Act, As per the current definitions, an MSME, in the case of an enterprise engaged in the manufacture or production of goods would be an enterprise where the investment in plant and machinery does not exceed INR 10 crores (approximately, USD 163,600) and in the case of an enterprise engaged in providing or rendering of services would be an enterprise where the investment in equipment does not exceed INR 5 crores (approximately USD 2
3 81,700). To support the claim to a small entity, the applicant is required to produce documentary proof accordingly. Some monetary concessions have been provided to applicants who fall under the category of small and medium size enterprises. The amendments have also led to a revised official fee schedule with a general substantial increase in fees. In an attempt to encourage applicants to use the electronic filing mode available on the Patent Office website, the amendment has introduced a 10% additional surcharge on manual mode of submitting documents under each activity, thereby making paper filing of documents a bit costlier than the electronic filing mode. II Recent development at the Patent Office 1) Indian Patent Office starts functioning as International Searching Authority (ISA) and International Preliminary Examining Authority (IPEA) On September 8, 2014, a new building with state of art facility was inaugurated for the functioning of the Indian Patent Office as International Searching Authority (ISA) and International Preliminary Examining Authority (IPEA) under the Patent Cooperation Treaty (PCT). Work has already started on this front and India has marked its name among the other ISA/IPEA recognized under the PCT. 2) Guidelines for examination of patent applications With a view to streamline the examination of patent applications and to give critical guidance to examiners for examining applications from various technical domains, the Patent Office has initiated issuance of guidelines for examiners. Till now, such guidelines have been issued and made public for Biotechnology related inventions as well as Traditional Knowledge and Biological Materials. In the last year, draft guidelines have been made available for examination of Computer Related Inventions (CRI) and Pharmaceutical 3
4 inventions. Stakeholder meetings have been held and the final versions will be issued shortly. III Patent Practice change based on case law 1) Proof of right mandatory for all patent applicants In an order passed in the matter of NTT DoCoMo Inc. Vs. The Controller of Patents & Design, the Intellectual Property Appellate Board (IPAB) has held that in cases where a patent applicant is not the true and first inventor of the invention, it is mandatory to file a proof of right while applying for the patent. This ruling is applicable to all patent applications, irrespective of whether they are being filed in India as Convention applications or as PCT National Phase applications. A notification issued by the Controller General of Patents in 2004 had waived the requirement to submit such proof in cases where the patent applicant was the same in the priority country and in India. Relying on this notification, the applicant in this case did not submit the proof of right document as it was the applicant in the convention country and in India. The IPAB, however, rejected this contention and held that since the law was clear in this respect, a proof of right must be furnished by applicant, irrespective of the application being filed through the Convention or the PCT National Phase route in India. In effect, the IPAB s order nullified the 2004 notification. The order is, however, silent as to whether this requirement is applicable only to cases which are currently pending or even those cases which have been granted in the last decade without the proof of right document on record. The IPAB appears to have overlooked the fact that proof of the right does not have to be only an assignment. Priority document as well as declaration of inventorship which are filed only when convention priority is claimed also may constitute valid proof of the right. IV Judicial pronouncements 1) The Supreme Court has held that a person cannot keep attacking a patent 4
5 In Dr. Aloys Wobben and Ors. vs. Yogesh Mehra and Ors. (Supreme Court, 2014), the Supreme Court of India has passed a landmark judgment retraining a person from taking multiple shots at a patent. In particular, the judgment laid down the following rules: If prior to the infringement suit, a defendant has filed revocation under Section 64(1) before the IPAB, then the defendant would be disentitled to file a counter claim; If the defendant counter claims for revocation in a suit, he cannot thereafter file revocation under Section 64(1) before the IPAB; and If any interested person has filed a post-grant opposition under Section 25(2), he is barred from filing revocation before the IPAB under Section 64(1) or filing a counter claim as a defendant in an infringement suit. 2) Mumbai High Court upholds compulsory license to NATCO In 2012, the Indian Patent Office granted a compulsory license in favour of Hyderabad based Natco Pharma Limited ( Natco ) for the anti-cancer drug, Sorafenib Tosylate, used for palliative treatment of liver and renal cancer. The drug is claimed by an Indian patent, being no that was granted to Bayer Corporation ( Bayer ) in The drug, sold by Bayer under the brand name Nexavar, was launched in India in Bayer appealed the Controller s order at the IPAB. IPAB ruled in favour of Natco, reaffirming the conclusion arrived at by the Controller General. The IPAB however clarified that import per se would qualify as working the patent within the scope of the Patents Act. However, the Patentee will be required to give reasons as to why the drug cannot be manufactured locally. Bayer had appealed before the Bombay High Court against the IPAB order which upheld the Compulsory License for Nexavar issued by the Indian 5
6 Patent Office to Natco. In a recent order, the Bombay High Court upheld the IPAB decision that the grant of compulsory licenses is to be considered on a case to case basis. Further, importation is considered as working so long as the patentee can justify the same. 3) Patent law emerging in telecom sector In a recent appeal [XU Dejun & Others vs. Vringo Infrastructure, Inc. & Another] concerning a CDMA technology patent, a Division Bench of the Delhi High Court, reversed the trend of setting interim arrangements in the form of royalty rates in favour of a plaintiff, where the plaintiff alleges infringement of Standard Essential Patents (SEPs) and claims FRAND (fair, reasonable and non-discriminatory) negotiations. Recognizing the time sensitivity of patent litigation matters, the Court, inter alia, ordered an expedited trial of the suit. In the early part of 2013, in the Ericsson vs. Micromax series of cases, the Delhi High Court had ordered Micromax to pay interim royalty to Ericsson during the pendency of the suit based on Ericsson s contention that Micromax had infringed its SEPs on 2G, 3G, EDGE and AMR technologies and claimed royalty on FRAND terms. In the backdrop of the above order, in November 2013, Vringo Inc. (along with its wholly owned subsidiary Vringo Infrastructure Inc.), a US based patent holding company, obtained an ex parte ad interim injunction order from the Delhi High Court in a suit for patent infringement against the Chinese telecom equipment giant, ZTE Corporation. Vide an order dated November 8, 2013, ZTE was restrained from manufacturing, assembling, importing, selling or offering for sale or advertising their products (telephone instruments, mobile handsets, tablets, handheld devices, dongles etc.) or any other similar devices that include the 3G technology CDMA2000 technology which may be covered by the said patent of Vringo Inc. Before the Single Judge of the Delhi High Court, Vringo had claimed that it acquired a patent portfolio including SEPs in the field of CDMA 6
7 technology from Nokia. The suit was for the infringement of one of such acquired patents (Indian Patent number ), claimed to be an SEP in the field of CDMA technology. Vringo alleged that since ZTE Corporation was manufacturing various CDMA2000 compliant products (such as mobile phones, tablets, dongles and other infrastructure equipment), it would necessarily require to use the technology which was the subject matter of the suit patent. Accordingly, Vringo alleged that ZTE could not import, sell or market the said products in India without obtaining a license from Vringo on FRAND terms. The Single Judge granted an ex parte interim order in favour of Vringo. Aggrieved by the said order, ZTE filed an appeal before a Division Bench of Delhi High Court. ZTE, inter alia, claimed non-infringement and contended that Vringo, being a non-practising entity (NPE) has not worked the patent in India. Vringo Infrastructure has sued ZTE globally in what appears to be an attempt to force ZTE to agree to FRAND licensing terms. ZTE took the categorical plea that FRAND licensing should be preceded by proof of validity of patent being established and the issue of infringement being held against ZTE. Relying on the view taken in the Ericsson v. Micromax orders, Vringo argued that ZTE be directed to first pay royalty on per product basis till the pendency of the suit. After consideration of the arguments of both the parties, the Court passed a consent order vacating the ex parte interim injunction, without fixing any royalty rates in favour of Vringo. Further, the Court declined to follow the precedent set in Ericson v. Micromax series of cases. The case is still in progress. 5) Interim injunctions granted in several pharmaceutical patent cases We also witnessed a trend of the High Court towards granting interim injunctions in pharmaceutical patent matters. 7
8 Novartis patent suits in Vildagliptin In a move to enforce its Indian Patent No for Type 2 Diabetes Mellitus (Type2DM) compound Vildagliptin, Novartis initiated a series of suits as quia timet action before the Delhi High Court against various pharmaceutical companies. The said patent was filed as a national phase application in India from PCT International Application No. PCT/EP99/09708 dated as a mail box application claiming priority from US Patent Application No. 09/209,068 dated Said patent was granted by the Indian Patent Office on 17/12/2007. Novartis is marketing Vildagliptin in India as two separate pharmaceutical products. While one contains Vildagliptin only as the Active Pharmaceutical Ingredient (API) under the brand name GALVUS, the other comprises Vildagliptin and Metformin Hydrochloride sold under the brand name GALVUSMET. Vildagliptin is believed to be the first gliptin developed, second being Sitagliptin (marketed by Merck under the brand name JANUVIA). Novartis had applied under Right to Information Act, 2005 (RTI) seeking information from the relevant State Drug Controlling authority regarding grant of permission to manufacture, sell and market Vildagliptin and Metformin Hydrochloride pharmaceutical products to entities in India. Based on the reply received from the respective State Drug Controlling authority, Novartis initiated infringement suits before the Delhi High Court against the entities who had been granted permission to manufacture and sell Vildagliptin and/or Metformin Hydrochloride pharmaceutical products. In all, Novartis has initiated quia timet action by way of infringement suit filed before Delhi High Court against 7 pharmaceutical companies namely Zee Laboratories Ltd., Wockhardt Ltd., Biocon Limited, Alembic Pharmaceuticals Ltd., Cadila Healthcare Ltd., Glenmark Generics Ltd. and Bajaj Healthcare Ltd. 8
9 Novartis main contention was that, since the Defendants had been granted the permission to manufacture Vildagliptin pharmaceutical products it was established that the Defendants intent to launch such product which will constitute infringement of the suit patent. Further, Novartis stated that as per their knowledge the Defendants have not yet launched their products in the market. Considering the arguments made on both sides, the High Court in the above suits favoured injunctions in favour of Novartis in some of the suits while passing restraining orders in others. 6) Positive news for bio-nutraceuticals In a well-reasoned order [Kibow Biotech Inc. vs. La Renon Healthcare Pvt. Ltd.] in November 2013, the IPAB set aside presumptions made about India s position on pharma/biopharma patents post the Supreme Court decision on Novartis s anticancer drug, Glivec. The IPAB upheld a product patent relating to a dietary supplement comprising a probiotic composition that reduced the need for kidney patients to undergo dialysis. The product patent entitled, Compositions for Augmenting Kidney Function was registered in the name of Kibow Biotech Inc., a US-based pharmaceutical company, which manufactured this product under the brand name Renadyl. The patent application was filed on March 28, 2006, for a probiotic composition for augmenting kidney function by removing toxic nitrogenous metabolic byproducts and inhibiting the overgrowth of undesirable bacteria in the gastrointestinal tract. The original patent application contained 20 claims, covering both product claims as well as method claims. However, when the patent was granted on September 29, 2009, these were eventually amended to 10 claims covering the composition, and the method claims were deleted as they were not permitted under Indian Patent law. In 2012, a revocation petition was filed against this patent by La Renon Healthcare Pvt Ltd., ( La Renon ) a Gujarat based Indian company, which sold a similar product in the market 9
10 under the brand Cudo. In 2011, the Madras High Court had dismissed a suit by Kibow Biotech against La Renon for infringement of its patents, including the patent under discussion here. The revocation petition claimed that the invention was not patentable under Indian law and raised grounds of obviousness and lack of inventive step. Though La Renon, had filed several documents in support of the ground of obviousness, the IPAB noted that 33 of such documents, were unsupported by any pleadings or grounds. As for the remaining prior art documents, the IPAB noted that none of the documents taught, either alone or in a combination, the specific limitations claimed by Kibow Biotech in its patent. While rejecting the ground of obviousness, the IPAB noted that mere speculation of a hypothesis was not enough to defeat the claimed invention on the ground of obviousness. The IPAB also rejected La Renon s argument that the composition claimed by Kibow Biotech was a mere admixture and therefore, did not qualify as patentable subject matter. It held that the disclosure in the specification of the patent went against La Renon s contention and that the claimed composition was indeed a synergistic composition having unexpected benefits and was not a mere admixture. Lastly, La Renon alleged false suggestion and misrepresentation in obtaining the patent, stating that the composition as claimed could not be administered to patients with renal problem because the various ingredients of the composition, such as, protein and fat soluble vitamins A, D, E and K would over-stress the kidney by increasing nitrogenous waste in the body. Kibow Biotech countered that the composition of the impugned patent did not claim to treat kidney disorder but acted in the intestine on the unfiltered waste and toxins present in blood, thereby enhancing the functions performed by the kidney during normal conditions. The IPAB agreed with Kibow Biotech that augmenting kidney function was not the same as augmenting the kidney functioning per se. It was, therefore, held that the petitioner had deliberately 10
11 misconstrued the scope and ambit of the invention. Based on the above analysis, the IPAB dismissed the revocation petition and imposed costs of INR 20,000 (approximately USD 320) on the petitioner, La Renon Healthcare Pvt. Ltd. This order passed by the IPAB is one of the few orders on cases involving biotech based products in India and has indeed been received very well by innovator companies in India and abroad. According to news reports, following the positive order from the IPAB, Kibow Biotech Inc. is all geared up to enter India s fast-growing dietary supplement market with its various proprietary products, including Renadyl. CONCLUSION While the statutory provisions have more or less remained the same, interpretation by both the IPAB and the Courts have been relatively progressive and liberal over the past year. 11
EMERGING IP RIGHTS. Country Report, India. D. Calab Gabriel
 RECENT DEVELOPMENT AND IMPORTANT DECISIONS IN INDIAN PATENT LAW ASIAN PATENT ATTORNEYS ASSOCIATION 63 rd Council Meeting Penang, Malaysia 8 th to 11 th November, 2014 EMERGING IP RIGHTS Country Report,
RECENT DEVELOPMENT AND IMPORTANT DECISIONS IN INDIAN PATENT LAW ASIAN PATENT ATTORNEYS ASSOCIATION 63 rd Council Meeting Penang, Malaysia 8 th to 11 th November, 2014 EMERGING IP RIGHTS Country Report,
Demystifying India s Patent Regime
 Demystifying India s Patent Regime Pankaj Soni October 30, 2014 www.remfry.com 1 AGENDA A Snapshot of India s Enforcement System Recent Decisions Impacting Patent Prosecution Proof of Right Section 8 Reporting
Demystifying India s Patent Regime Pankaj Soni October 30, 2014 www.remfry.com 1 AGENDA A Snapshot of India s Enforcement System Recent Decisions Impacting Patent Prosecution Proof of Right Section 8 Reporting
Ritushka Negi Remfry & Sagar, Partner
 Ritushka Negi Remfry & Sagar, Partner NO HOLDS BARRED KEY TAKEAWAYS FROM RECENT PATENT DECISIONS IN INDIA RITUSHKA NEGI November 21, 2016 www.remfry.com 3 Administrative & Judicial Hierarchy Supreme Court
Ritushka Negi Remfry & Sagar, Partner NO HOLDS BARRED KEY TAKEAWAYS FROM RECENT PATENT DECISIONS IN INDIA RITUSHKA NEGI November 21, 2016 www.remfry.com 3 Administrative & Judicial Hierarchy Supreme Court
: 1 : Time allowed : 3 hours Maximum marks : 100. Total number of questions : 6 Total number of printed pages : 7
 OPEN BOOK EXAMINATION Roll No : 1 : NEW SYLLABUS Time allowed : 3 hours Maximum marks : 100 Total number of questions : 6 Total number of printed pages : 7 NOTE : Answer ALL Questions. 1. Read the case
OPEN BOOK EXAMINATION Roll No : 1 : NEW SYLLABUS Time allowed : 3 hours Maximum marks : 100 Total number of questions : 6 Total number of printed pages : 7 NOTE : Answer ALL Questions. 1. Read the case
4. COMPARISON OF THE INDIAN PATENT LAW WITH THE PATENT LAWS IN U.S., EUROPE AND CHINA
 4. COMPARISON OF THE INDIAN PATENT LAW WITH THE PATENT LAWS IN U.S., EUROPE AND CHINA Provisions of the Indian patent law were compared with the relevant provisions of the patent laws in U.S., Europe and
4. COMPARISON OF THE INDIAN PATENT LAW WITH THE PATENT LAWS IN U.S., EUROPE AND CHINA Provisions of the Indian patent law were compared with the relevant provisions of the patent laws in U.S., Europe and
Merck Sharp & Dohme & Anr. v Glenmark Pharmaceuticals Ltd
 BIOTECH BUZZ International Subcommittee December 2015 Contributor: Archana Shanker Changing trends in Indian patent enforcement In the history of the Patent Litigation in India, at least since 1970, only
BIOTECH BUZZ International Subcommittee December 2015 Contributor: Archana Shanker Changing trends in Indian patent enforcement In the history of the Patent Litigation in India, at least since 1970, only
LEGAL DEVELOPMENTS. The important legal updates from the previous quarter are summarized below: Trade Marks Rules, 2017 Notified
 z This Newsletter brings to you the IP updates during the first quarter of this year. The first quarter saw remarkable changes in trademark practice and procedure in India. With substantial changes in
z This Newsletter brings to you the IP updates during the first quarter of this year. The first quarter saw remarkable changes in trademark practice and procedure in India. With substantial changes in
ASIAN PATENT ATTORNEYS ASSOCIATION. 62 nd Council Meeting. Hanoi, Vietnam. Patent Committee Report: INDIA. Hari Subramaniam, Neeti Dewan, Sanjay Kumar
 ASIAN PATENT ATTORNEYS ASSOCIATION 62 nd Council Meeting Hanoi, Vietnam Patent Committee Report: INDIA Hari Subramaniam, Neeti Dewan, Sanjay Kumar 1 India: Patents 2013 There have been no changes in statutory
ASIAN PATENT ATTORNEYS ASSOCIATION 62 nd Council Meeting Hanoi, Vietnam Patent Committee Report: INDIA Hari Subramaniam, Neeti Dewan, Sanjay Kumar 1 India: Patents 2013 There have been no changes in statutory
October-November 2013 Edition Click to read in detail
 Newsletter October-November 2013 Edition Click to read in detail India starts functioning as International Searching Authority Dynamic Trade Marks Utilities portal launched Dasatinib compulsory licence
Newsletter October-November 2013 Edition Click to read in detail India starts functioning as International Searching Authority Dynamic Trade Marks Utilities portal launched Dasatinib compulsory licence
Examining Patent Enforcement and Litigation in India from A Development Perspective A study
 Examining Patent Enforcement and Litigation in India from A Development Perspective A study Ayyappan Palanissamy + School of Business and Design, Swinburne University of Technology Sarawak, Kuching, Malaysia
Examining Patent Enforcement and Litigation in India from A Development Perspective A study Ayyappan Palanissamy + School of Business and Design, Swinburne University of Technology Sarawak, Kuching, Malaysia
Prathiba M. Singh President, APAA (Indian Group)
 Prathiba M. Singh President, APAA (Indian Group) Section 108 relates to relief in a suit for infringement Section 108(1) provides for Damages or Account of Profits At the option of the Plaintiff Section
Prathiba M. Singh President, APAA (Indian Group) Section 108 relates to relief in a suit for infringement Section 108(1) provides for Damages or Account of Profits At the option of the Plaintiff Section
Bajaj Auto Ltd vs. TVS Motor Company Ltd
 Bajaj Auto Ltd vs. TVS Motor Company Ltd Case History July 16,2002 Bajaj files patent application October 30,2003 Bajaj files international patent applications in foreign countries July 7,2005 Bajaj granted
Bajaj Auto Ltd vs. TVS Motor Company Ltd Case History July 16,2002 Bajaj files patent application October 30,2003 Bajaj files international patent applications in foreign countries July 7,2005 Bajaj granted
Patent Enforcement in India
 Patent Enforcement in India Intellectual property assets are touted as the cornerstone of competitiveness in international trade and are the driving factors behind socio-economic development in India.
Patent Enforcement in India Intellectual property assets are touted as the cornerstone of competitiveness in international trade and are the driving factors behind socio-economic development in India.
IN THE HIGH COURT OF DELHI AT NEW DELHI SUBJECT : SUIT FOR INJUNCTION Date of decision: 5th April, CS(OS) 586/2013
 IN THE HIGH COURT OF DELHI AT NEW DELHI SUBJECT : SUIT FOR INJUNCTION Date of decision: 5th April, 2013. CS(OS) 586/2013 MERCK SHARP AND DOHME CORPORATION & ANR...Plaintiffs Through: Mr. Parag P. Tripathi,
IN THE HIGH COURT OF DELHI AT NEW DELHI SUBJECT : SUIT FOR INJUNCTION Date of decision: 5th April, 2013. CS(OS) 586/2013 MERCK SHARP AND DOHME CORPORATION & ANR...Plaintiffs Through: Mr. Parag P. Tripathi,
Time allowed : 3 hours Maximum marks : 100. Total number of questions : 6 Total number of printed pages : 8
 OPEN BOOK EXAMINATION Roll No... : 1 : 344 Time allowed : 3 hours Maximum marks : 100 Total number of questions : 6 Total number of printed pages : 8 NOTE : Answer ALL Questions. 1. Read the following
OPEN BOOK EXAMINATION Roll No... : 1 : 344 Time allowed : 3 hours Maximum marks : 100 Total number of questions : 6 Total number of printed pages : 8 NOTE : Answer ALL Questions. 1. Read the following
* IN THE HIGH COURT OF DELHI AT NEW DELHI. versus CORAM: HON'BLE MR. JUSTICE JAYANT NATH
 * IN THE HIGH COURT OF DELHI AT NEW DELHI Decided on : April 25, 2014 + IA No. 5745/2013 (u/o 39 R 1 & 2 CPC) in CS(OS) 660/2013 WOCKHARDT LTD. Through... Plaintiff Mr.Ajay Sahni, Ms. Kanika Bajaj and
* IN THE HIGH COURT OF DELHI AT NEW DELHI Decided on : April 25, 2014 + IA No. 5745/2013 (u/o 39 R 1 & 2 CPC) in CS(OS) 660/2013 WOCKHARDT LTD. Through... Plaintiff Mr.Ajay Sahni, Ms. Kanika Bajaj and
INDIAN PATENTS. Request for Examination. 48 months from priority*
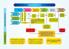 INDIAN PATENTS INDIAN PATENT PROSECUTION ASA FACILITATION Direct filing (Priority application) 31 months from Priority (PCT Route) Filing 12 months from Basic Application (Convention Route) 18 months from
INDIAN PATENTS INDIAN PATENT PROSECUTION ASA FACILITATION Direct filing (Priority application) 31 months from Priority (PCT Route) Filing 12 months from Basic Application (Convention Route) 18 months from
The India Patent System: A Decade in Review
 Cybaris Volume 8 Issue 1 Article 2 2017 The India Patent System: A Decade in Review Vindhya S. Mani Divyanshu Srivastava Mukundan Chakrapani Jay Erstling Follow this and additional works at: http://open.mitchellhamline.edu/cybaris
Cybaris Volume 8 Issue 1 Article 2 2017 The India Patent System: A Decade in Review Vindhya S. Mani Divyanshu Srivastava Mukundan Chakrapani Jay Erstling Follow this and additional works at: http://open.mitchellhamline.edu/cybaris
The Judgment can be accessed here at the website of the Delhi High Court. The Judgment can also be accessed here at India Kanoon website.
 The Judgment can be accessed here at the website of the Delhi High Court. The Judgment can also be accessed here at India Kanoon website. The Facts: The brief facts of the case are as follows: The Plaintiff
The Judgment can be accessed here at the website of the Delhi High Court. The Judgment can also be accessed here at India Kanoon website. The Facts: The brief facts of the case are as follows: The Plaintiff
THE PATENTS NEWSLETTER
 THE PATENTS NEWSLETTER In the whirlwind of events marking the patent landscape in India, we strive to keep you updated with our regular editions of the Patents Newsletter and bring you patents news from
THE PATENTS NEWSLETTER In the whirlwind of events marking the patent landscape in India, we strive to keep you updated with our regular editions of the Patents Newsletter and bring you patents news from
US-China Business Council Comments on the Draft Measures for the Compulsory Licensing of Patents
 US-China Business Council Comments on the Draft Measures for the Compulsory Licensing of Patents The US-China Business Council (USCBC) and its member companies appreciate the opportunity to submit comments
US-China Business Council Comments on the Draft Measures for the Compulsory Licensing of Patents The US-China Business Council (USCBC) and its member companies appreciate the opportunity to submit comments
RASHTRASANT TUKADOJI MAHARAJ NAGPUR UNIVERSITY S. Dr. BABASAHEB AMBEDKAR COLLEGE OF LAW, NAGPUR MOOT PROBLEM 1
 RASHTRASANT TUKADOJI MAHARAJ NAGPUR UNIVERSITY S Dr. BABASAHEB AMBEDKAR COLLEGE OF LAW, NAGPUR 14 th JUSTA CAUSA NATIONAL LAW FESTIVAL MOOT PROBLEM 1 Ram Rahim Joseph (RRJ) is a right handed Indian Citizen
RASHTRASANT TUKADOJI MAHARAJ NAGPUR UNIVERSITY S Dr. BABASAHEB AMBEDKAR COLLEGE OF LAW, NAGPUR 14 th JUSTA CAUSA NATIONAL LAW FESTIVAL MOOT PROBLEM 1 Ram Rahim Joseph (RRJ) is a right handed Indian Citizen
Intellectual Property Rights Relating to Trademarks, Copyrights, Patents and Designs
 Intellectual Property Rights Relating to Trademarks, Copyrights, Patents and Designs National Judicial Academy, Bhopal Pravin Anand 24 th September, 2016 BACKGROUND Almost 20 International Treaties concerning
Intellectual Property Rights Relating to Trademarks, Copyrights, Patents and Designs National Judicial Academy, Bhopal Pravin Anand 24 th September, 2016 BACKGROUND Almost 20 International Treaties concerning
* IN THE HIGH COURT OF DELHI AT NEW DELHI. % Order delivered on: 2 nd September, I.A. No.17351/2015 in CS (OS) 2501/2015.
 * IN THE HIGH COURT OF DELHI AT NEW DELHI % Order delivered on: 2 nd September, 2015 + I.A. No.17351/2015 in CS (OS) 2501/2015 TELEFONAKTIEBOLAGET LM ERICSSON (PUBL)... Plaintiff Through Mr. C.S.Vaidyanathan,
* IN THE HIGH COURT OF DELHI AT NEW DELHI % Order delivered on: 2 nd September, 2015 + I.A. No.17351/2015 in CS (OS) 2501/2015 TELEFONAKTIEBOLAGET LM ERICSSON (PUBL)... Plaintiff Through Mr. C.S.Vaidyanathan,
DELHI HIGH COURT UPHELD JUDGMENT DIRECTING RESTORATION AND RENEWAL OF TRADEMARK MBD, 29 YEARS AFTER DUE DATE OF RENEWAL
 From the SelectedWorks of Sudhir Kumar Aswal Summer March 11, 2013 DELHI HIGH COURT UPHELD JUDGMENT DIRECTING RESTORATION AND RENEWAL OF TRADEMARK MBD, 29 YEARS AFTER DUE DATE OF RENEWAL Sudhir Kumar Aswal
From the SelectedWorks of Sudhir Kumar Aswal Summer March 11, 2013 DELHI HIGH COURT UPHELD JUDGMENT DIRECTING RESTORATION AND RENEWAL OF TRADEMARK MBD, 29 YEARS AFTER DUE DATE OF RENEWAL Sudhir Kumar Aswal
PROFESSIONAL PROGRAMME UPDATES FOR INTELLECTUAL PROPERTY RIGHTS: LAWS AND PRACTICES MODULE 3- ELECTIVE PAPER 9.4
 PROFESSIONAL PROGRAMME UPDATES FOR INTELLECTUAL PROPERTY RIGHTS: LAWS AND PRACTICES (Relevant for students appearing in December, 2017 examination) MODULE 3- ELECTIVE PAPER 9.4 Disclaimer: This document
PROFESSIONAL PROGRAMME UPDATES FOR INTELLECTUAL PROPERTY RIGHTS: LAWS AND PRACTICES (Relevant for students appearing in December, 2017 examination) MODULE 3- ELECTIVE PAPER 9.4 Disclaimer: This document
ASN 1/52 WP-1323-Jud. IN THE HIGH COURT OF JUDICATURE AT BOMBAY ORDINARY ORIGINAL CIVIL JURISDICTION
 ASN 1/52 WP-1323-Jud. IN THE HIGH COURT OF JUDICATURE AT BOMBAY ORDINARY ORIGINAL CIVIL JURISDICTION WRIT PETITION NO.1323 OF 2013 Bayer Corporation A Corporation Organized under the laws of the State
ASN 1/52 WP-1323-Jud. IN THE HIGH COURT OF JUDICATURE AT BOMBAY ORDINARY ORIGINAL CIVIL JURISDICTION WRIT PETITION NO.1323 OF 2013 Bayer Corporation A Corporation Organized under the laws of the State
The World Intellectual Property Organization
 The World Intellectual Property Organization The World Intellectual Property Organization is an international organization dedicated to ensuring that the rights of creators and owners of intellectual property
The World Intellectual Property Organization The World Intellectual Property Organization is an international organization dedicated to ensuring that the rights of creators and owners of intellectual property
BE it enacted by Parliament in the Fifty-sixth Year of the Republic of India as follows:-
 ~ THE PATENTS (AMENDMENT) ACT, 2005 # NO. 15 OF 2005 $ [4th April, 2005] + An Act further to amend the Patents Act, 1970. BE it enacted by Parliament in the Fifty-sixth Year of the Republic of India as
~ THE PATENTS (AMENDMENT) ACT, 2005 # NO. 15 OF 2005 $ [4th April, 2005] + An Act further to amend the Patents Act, 1970. BE it enacted by Parliament in the Fifty-sixth Year of the Republic of India as
$~ * IN THE HIGH COURT OF DELHI AT NEW DELHI
 $~ * IN THE HIGH COURT OF DELHI AT NEW DELHI + CS(COMM) 1290/2016 THE COCA-COLA COMPANY & ANR... Plaintiffs Through: Mr Karan Bajaj with Ms Kripa Pandit and Mr Dhruv Nayar, Advocates versus GLACIER WATER
$~ * IN THE HIGH COURT OF DELHI AT NEW DELHI + CS(COMM) 1290/2016 THE COCA-COLA COMPANY & ANR... Plaintiffs Through: Mr Karan Bajaj with Ms Kripa Pandit and Mr Dhruv Nayar, Advocates versus GLACIER WATER
ip LAB Delhi High Court frees Indian drug regulator from the shackles of patent linkage Nishith Desai Associates Legal & Tax Counseling Worldwide
 ip LAB Delhi High Court frees Indian drug regulator from the shackles of patent linkage IP Pharma Team Aditi Nadkarni & Gowree Gokhale October 26, 2009 This ip Lab is a copyright of Nishith Desai Associates.
ip LAB Delhi High Court frees Indian drug regulator from the shackles of patent linkage IP Pharma Team Aditi Nadkarni & Gowree Gokhale October 26, 2009 This ip Lab is a copyright of Nishith Desai Associates.
The patent opposition process
 The patent opposition process Interested parties can use the two-stage opposition procedure to challenge a patent either pre or post-grant a broad window in which to take action By G Deepak Sriniwas and
The patent opposition process Interested parties can use the two-stage opposition procedure to challenge a patent either pre or post-grant a broad window in which to take action By G Deepak Sriniwas and
The Patents (Amendment) Act,
 !"# The Patents (Amendment) Act, 2005 1 [NO. 15 OF 2005] CONTENTS [April 4, 2005] Sections Sections 1. Short title and commencement 40. Amendment of Section 57 2. Amendment of Section 2 41. Substitution
!"# The Patents (Amendment) Act, 2005 1 [NO. 15 OF 2005] CONTENTS [April 4, 2005] Sections Sections 1. Short title and commencement 40. Amendment of Section 57 2. Amendment of Section 2 41. Substitution
M/s. BDR Pharmaceuticals International Pvt. Ltd Applicant VERSUS
 BDR Pharmaceuticals International Pvt. Ltd v. Bristol Myers Squibb Company Controller of Patents, Patents Office, Mumbai (BEFORE CHAITANYA PRASAD, CONTROLLER) M/s. BDR Pharmaceuticals International Pvt.
BDR Pharmaceuticals International Pvt. Ltd v. Bristol Myers Squibb Company Controller of Patents, Patents Office, Mumbai (BEFORE CHAITANYA PRASAD, CONTROLLER) M/s. BDR Pharmaceuticals International Pvt.
IN THE HIGH COURT OF DELHI AT NEW DELHI. CS (OS) No of Versus CORAM: JUSTICE S. MURALIDHAR O R D E R
 IN THE HIGH COURT OF DELHI AT NEW DELHI CS (OS) No. 2206 of 2012 KONINKLIJKE PHILIPS ELECTRONICS N.V.... Plaintiff Through: Mr. Sudhir Chandra, Senior Advocate with Mr. Pravin Anand, Ms. Vaishali Mittal,
IN THE HIGH COURT OF DELHI AT NEW DELHI CS (OS) No. 2206 of 2012 KONINKLIJKE PHILIPS ELECTRONICS N.V.... Plaintiff Through: Mr. Sudhir Chandra, Senior Advocate with Mr. Pravin Anand, Ms. Vaishali Mittal,
Before: MR. JUSTICE BIRSS Between: VRINGO INFRASTRUCTURE, INC.
 Neutral Citation Number: [2015] EWHC 1704 (Pat) IN THE HIGH COURT OF JUSTICE CHANCERY DIVISION PATENTS COURT Case No: HC-2012-000076 The Rolls Building 7 Rolls Buildings London EC4A 1NL Date: 08/06/2015
Neutral Citation Number: [2015] EWHC 1704 (Pat) IN THE HIGH COURT OF JUSTICE CHANCERY DIVISION PATENTS COURT Case No: HC-2012-000076 The Rolls Building 7 Rolls Buildings London EC4A 1NL Date: 08/06/2015
PROFESSIONAL PROGRAMME UPDATES FOR INTELLECTUAL PROPERTY RIGHTS: LAWS AND PRACTICES MODULE 3- ELECTIVE PAPER 9.4
 PROFESSIONAL PROGRAMME UPDATES FOR INTELLECTUAL PROPERTY RIGHTS: LAWS AND PRACTICES (Relevant for students appearing in December, 2018 examination) MODULE 3- ELECTIVE PAPER 9.4 Disclaimer: This document
PROFESSIONAL PROGRAMME UPDATES FOR INTELLECTUAL PROPERTY RIGHTS: LAWS AND PRACTICES (Relevant for students appearing in December, 2018 examination) MODULE 3- ELECTIVE PAPER 9.4 Disclaimer: This document
Attachment: Opinions on the Draft Amendment of the Implementing Regulations of the Patent Law of the People s Republic of China
 March 31, 2009 To: Legislative Affairs Office State Council People s Republic of China Hirohiko Usui President Japan Intellectual Property Association Opinions on the Draft Amendment of the Implementing
March 31, 2009 To: Legislative Affairs Office State Council People s Republic of China Hirohiko Usui President Japan Intellectual Property Association Opinions on the Draft Amendment of the Implementing
Patent Litigation in Taiwan: overview
 Patent Litigation in Taiwan: overview Resource type: Country Q&A Status: Law stated as at 01-Jan-2016 Jurisdiction: Taiwan A Q&A guide to patent litigation in Taiwan. The Q&A gives a high level overview
Patent Litigation in Taiwan: overview Resource type: Country Q&A Status: Law stated as at 01-Jan-2016 Jurisdiction: Taiwan A Q&A guide to patent litigation in Taiwan. The Q&A gives a high level overview
How patents work An introduction for law students
 How patents work An introduction for law students 1 Learning goals The learning goals of this lecture are to understand: the different types of intellectual property rights available the role of the patent
How patents work An introduction for law students 1 Learning goals The learning goals of this lecture are to understand: the different types of intellectual property rights available the role of the patent
$~28 * IN THE HIGH COURT OF DELHI AT NEW DELHI. versus. % Date of Decision: 06 th November, 2017 J U D G M E N T
 $~28 * IN THE HIGH COURT OF DELHI AT NEW DELHI + CS(COMM) 563/2017 MANKIND PHARMA LIMITED... Plaintiff Through: Ms.Ishanki Gupta with Mr.Harsh Vardhan, Advocates. versus SHAM LAL & ORS Through: None...
$~28 * IN THE HIGH COURT OF DELHI AT NEW DELHI + CS(COMM) 563/2017 MANKIND PHARMA LIMITED... Plaintiff Through: Ms.Ishanki Gupta with Mr.Harsh Vardhan, Advocates. versus SHAM LAL & ORS Through: None...
KSR & Co Company Secretaries LLP PRACTISING COMPANY SECRETARIES & TRADE MARK AGENTS COIMBATORE & CHENNAI
 KSR & Co Company Secretaries LLP PRACTISING COMPANY SECRETARIES & TRADE MARK AGENTS COIMBATORE & CHENNAI Assuring Assuring Compliances Compliances & Solutions & Solutions Beyond Beyond Challenge Challenge
KSR & Co Company Secretaries LLP PRACTISING COMPANY SECRETARIES & TRADE MARK AGENTS COIMBATORE & CHENNAI Assuring Assuring Compliances Compliances & Solutions & Solutions Beyond Beyond Challenge Challenge
THE PATENTS ACT 1970
 THE PATENTS ACT 1970 (39 of 1970) An Act to amend and consolidate the law relating to patents. (19 th September, 1970) Be it enacted by Parliament in the twenty first year of the Republic of India as follows;-
THE PATENTS ACT 1970 (39 of 1970) An Act to amend and consolidate the law relating to patents. (19 th September, 1970) Be it enacted by Parliament in the twenty first year of the Republic of India as follows;-
Rejected in India: Dr. Feroz Ali, Dr. Sudarsan Rajagopal, Mohamed Mustafa and Chinnasamy Prabhu WHAT THE INDIAN PATENT OFFICE GOT
 Rejected in India: WHAT THE INDIAN PATENT OFFICE GOT RIGHT ON PHARMACEUTICALS PATENT APPLICATIONS (2009 2016) Dr. Feroz Ali, Dr. Sudarsan Rajagopal, Mohamed Mustafa and Chinnasamy Prabhu December 2017
Rejected in India: WHAT THE INDIAN PATENT OFFICE GOT RIGHT ON PHARMACEUTICALS PATENT APPLICATIONS (2009 2016) Dr. Feroz Ali, Dr. Sudarsan Rajagopal, Mohamed Mustafa and Chinnasamy Prabhu December 2017
Remedies: Injunction and Damages. 1. General
 VI. Remedies: Injunction and Damages 1. General If infringement is found and validity of the patent is not denied by the court, then the patentee is entitled to the remedies of both injunction and damages
VI. Remedies: Injunction and Damages 1. General If infringement is found and validity of the patent is not denied by the court, then the patentee is entitled to the remedies of both injunction and damages
Exceptions and Limitation of Patent Rights and its Enforcement in India
 Journal of Intellectual Property Rights Vol 20, September 2015, pp 297-304 Exceptions and Limitation of Patent Rights and its Enforcement in India Biplab Kumar Lenin 1 and Harsha Rohatgi 2 1 IIT Kharagpur,
Journal of Intellectual Property Rights Vol 20, September 2015, pp 297-304 Exceptions and Limitation of Patent Rights and its Enforcement in India Biplab Kumar Lenin 1 and Harsha Rohatgi 2 1 IIT Kharagpur,
Second medical use or indication claims. [Please insert name last name in CAPITAL letters please]
![Second medical use or indication claims. [Please insert name last name in CAPITAL letters please] Second medical use or indication claims. [Please insert name last name in CAPITAL letters please]](/thumbs/82/85618671.jpg) Question Q238 National Group: Title: Contributors: Reporter within Working Committee: New Zealand Second medical use or indication claims Michael BROWN, Partner Helen BELLCHAMBERS, Associate A J Park [Please
Question Q238 National Group: Title: Contributors: Reporter within Working Committee: New Zealand Second medical use or indication claims Michael BROWN, Partner Helen BELLCHAMBERS, Associate A J Park [Please
Case 1:17-cv UNA Document 1 Filed 12/22/17 Page 1 of 10 PageID #: 1 IN THE UNITED STATES DISTRICT COURT FOR THE DISTRICT OF DELAWARE
 Case 1:17-cv-01844-UNA Document 1 Filed 12/22/17 Page 1 of 10 PageID #: 1 IN THE UNITED STATES DISTRICT COURT FOR THE DISTRICT OF DELAWARE AMGEN INC., v. Plaintiff, TORRENT PHARMACEUTICALS LTD. and TORRENT
Case 1:17-cv-01844-UNA Document 1 Filed 12/22/17 Page 1 of 10 PageID #: 1 IN THE UNITED STATES DISTRICT COURT FOR THE DISTRICT OF DELAWARE AMGEN INC., v. Plaintiff, TORRENT PHARMACEUTICALS LTD. and TORRENT
F-19 $~ * IN THE HIGH COURT OF DELHI AT NEW DELHI. MANKIND PHARMA LIMITED... Plaintiff Through: Ms. Ishanki Gupta, Advocate. versus.
 F-19 $~ * IN THE HIGH COURT OF DELHI AT NEW DELHI + CS(OS) 2982/2015 MANKIND PHARMA LIMITED... Plaintiff Through: Ms. Ishanki Gupta, Advocate. versus SUDHANSHU KUMAR & ANR. Through: None... Defendants
F-19 $~ * IN THE HIGH COURT OF DELHI AT NEW DELHI + CS(OS) 2982/2015 MANKIND PHARMA LIMITED... Plaintiff Through: Ms. Ishanki Gupta, Advocate. versus SUDHANSHU KUMAR & ANR. Through: None... Defendants
IN THE HIGH COURT OF DELHI AT NEW DELHI FAO (OS) 188/2008 F.HOFFMANN-LA ROCHE LTD. & ANR
 IN THE HIGH COURT OF DELHI AT NEW DELHI FAO (OS) 188/2008 F.HOFFMANN-LA ROCHE LTD. & ANR versus Date of decision: April 24 th 2009... Appellants Through Dr. A.M. Singhvi, Senior Advocate, Mr. Parag. P.
IN THE HIGH COURT OF DELHI AT NEW DELHI FAO (OS) 188/2008 F.HOFFMANN-LA ROCHE LTD. & ANR versus Date of decision: April 24 th 2009... Appellants Through Dr. A.M. Singhvi, Senior Advocate, Mr. Parag. P.
* IN THE HIGH COURT OF DELHI AT NEW DELHI
 * IN THE HIGH COURT OF DELHI AT NEW DELHI 32. + W.P.(C) No. 332 of 2010 M/S UCB FARCHIM SA... Petitioner Through: Mr. Sudhir Chandra, Sr. Advocate with Mr. Sanjay Kumar, Ms. Arpita Sawhney and Mr. Sukhdev,
* IN THE HIGH COURT OF DELHI AT NEW DELHI 32. + W.P.(C) No. 332 of 2010 M/S UCB FARCHIM SA... Petitioner Through: Mr. Sudhir Chandra, Sr. Advocate with Mr. Sanjay Kumar, Ms. Arpita Sawhney and Mr. Sukhdev,
IN THE HIGH COURT OF DELHI AT NEW DELHI FAO (OS) 367/2007. Date of Decision : 08 TH FEBRUARY, 2008
 IN THE HIGH COURT OF DELHI AT NEW DELHI SUBJECT : Code of Civil Procedure FAO (OS) 367/2007 Date of Decision : 08 TH FEBRUARY, 2008 EUREKA FORBES LTD. & ANR.... Appellants Through : Mr. Valmiki Mehta,
IN THE HIGH COURT OF DELHI AT NEW DELHI SUBJECT : Code of Civil Procedure FAO (OS) 367/2007 Date of Decision : 08 TH FEBRUARY, 2008 EUREKA FORBES LTD. & ANR.... Appellants Through : Mr. Valmiki Mehta,
versus CORAM: JUSTICE S. MURALIDHAR O R D E R IA No of 2011 (by Defendant u/o VII R. 10 & 11 CPC)
 IN THE HIGH COURT OF DELHI AT NEW DELHI CS (OS) 1188 of 2011 & IAs 7950 of 2011 (u/o 39 R. 1 & 2 CPC), 3388 of 2013 (u/o XXVI R. 2 CPC) & 18427 of 2013 (by Plaintiff u/o VII R. 14 CPC) LT FOODS LIMITED...
IN THE HIGH COURT OF DELHI AT NEW DELHI CS (OS) 1188 of 2011 & IAs 7950 of 2011 (u/o 39 R. 1 & 2 CPC), 3388 of 2013 (u/o XXVI R. 2 CPC) & 18427 of 2013 (by Plaintiff u/o VII R. 14 CPC) LT FOODS LIMITED...
TABLE OF CONTENTS CHAPTER 1 PATENTS AND UTILITY MODEL RIGHT 3
 TABLE OF CONTENTS TABLE OF CONTENTS CHAPTER 1 PATENTS AND UTILITY MODEL RIGHT 3 Ⅰ. Patents 3 1. Subjective requirements 3 2. Objective requirements 3 3. Procedural requirements 4 Ⅱ. Utility model right
TABLE OF CONTENTS TABLE OF CONTENTS CHAPTER 1 PATENTS AND UTILITY MODEL RIGHT 3 Ⅰ. Patents 3 1. Subjective requirements 3 2. Objective requirements 3 3. Procedural requirements 4 Ⅱ. Utility model right
Taiwan International Patent & Law Office
 HIGHLIGHTS ON THE PROPOSED PATENT ACT AMENDMENT OF TAIWAN AND COPYRIGHT LAW AMENDMENT As of November 2009, the proposed amendments to Taiwan s Patent Act are pending the final review and approval of the
HIGHLIGHTS ON THE PROPOSED PATENT ACT AMENDMENT OF TAIWAN AND COPYRIGHT LAW AMENDMENT As of November 2009, the proposed amendments to Taiwan s Patent Act are pending the final review and approval of the
ETHIOPIA A PROCLAMATION CONCERNING INVENTIONS, MINOR INVENTIONS AND INDUSTRIAL DESIGNS PROCLAMATION NO. 123/1995 ENTRY INTO FORCE: May 10, 1995
 ETHIOPIA A PROCLAMATION CONCERNING INVENTIONS, MINOR INVENTIONS AND INDUSTRIAL DESIGNS PROCLAMATION NO. 123/1995 ENTRY INTO FORCE: May 10, 1995 TABLE OF CONTENTS CHAPTER ONE General Provisions 1. Short
ETHIOPIA A PROCLAMATION CONCERNING INVENTIONS, MINOR INVENTIONS AND INDUSTRIAL DESIGNS PROCLAMATION NO. 123/1995 ENTRY INTO FORCE: May 10, 1995 TABLE OF CONTENTS CHAPTER ONE General Provisions 1. Short
* IN THE HIGH COURT OF DELHI AT NEW DELHI. + I.A. No.23086/2012 in CS(OS) No.3534/2012 ABBOTT HEALTHCARE PVT. LTD. versus
 * IN THE HIGH COURT OF DELHI AT NEW DELHI + I.A. No.23086/2012 in CS(OS) No.3534/2012 ABBOTT HEALTHCARE PVT. LTD. Through versus RAJ KUMAR PRASAD & ORS. Decided on :25.04.2014...Plaintiff Mr.Manav Kumar,
* IN THE HIGH COURT OF DELHI AT NEW DELHI + I.A. No.23086/2012 in CS(OS) No.3534/2012 ABBOTT HEALTHCARE PVT. LTD. Through versus RAJ KUMAR PRASAD & ORS. Decided on :25.04.2014...Plaintiff Mr.Manav Kumar,
Patent Act, B.E (1979) As Amended until Patent Act (No.3), B.E (1999) Translation
 Patent Act, B.E. 2522 (1979) As Amended until Patent Act (No.3), B.E. 2542 (1999) Translation BHUMIBOL ADULYADEJ, REX. Given on the 11th day of March, B.E. 2522; Being the 34th year of the present Reign
Patent Act, B.E. 2522 (1979) As Amended until Patent Act (No.3), B.E. 2542 (1999) Translation BHUMIBOL ADULYADEJ, REX. Given on the 11th day of March, B.E. 2522; Being the 34th year of the present Reign
IP CONCLAVE 2010, MUMBAI STRATEGIES WITH US PATENT PRACTICE NAREN THAPPETA US PATENT ATTORNEY & INDIA PATENT AGENT BANGALORE, INDIA
 IP CONCLAVE 2010, MUMBAI STRATEGIES WITH US PATENT PRACTICE NAREN THAPPETA US PATENT ATTORNEY & INDIA PATENT AGENT BANGALORE, INDIA www.iphorizons.com Not legal Advise! Broad Organization A. Pre filing
IP CONCLAVE 2010, MUMBAI STRATEGIES WITH US PATENT PRACTICE NAREN THAPPETA US PATENT ATTORNEY & INDIA PATENT AGENT BANGALORE, INDIA www.iphorizons.com Not legal Advise! Broad Organization A. Pre filing
* IN THE HIGH COURT OF DELHI AT NEW DELHI % Reserved on: 12 th March, 2018 Pronounced on: 12 th April, 2018 CORAM: HON'BLE MR. JUSTICE YOGESH KHANNA
 * IN THE HIGH COURT OF DELHI AT NEW DELHI % Reserved on: 12 th March, 2018 Pronounced on: 12 th April, 2018 + CS(COMM) 712/2018 VIOR(INTERNATIONAL) LTD & ANR Through : versus MAXYCON HEALTH CARE PRIVATE
* IN THE HIGH COURT OF DELHI AT NEW DELHI % Reserved on: 12 th March, 2018 Pronounced on: 12 th April, 2018 + CS(COMM) 712/2018 VIOR(INTERNATIONAL) LTD & ANR Through : versus MAXYCON HEALTH CARE PRIVATE
WIPO WORLD INTELLECTUAL PROPERTY ORGANISATION ARBITRATION RULES
 APPENDIX 3.17 WIPO WORLD INTELLECTUAL PROPERTY ORGANISATION ARBITRATION RULES (as from 1 October 2002) I. GENERAL PROVISIONS Abbreviated Expressions Article 1 In these Rules: Arbitration Agreement means
APPENDIX 3.17 WIPO WORLD INTELLECTUAL PROPERTY ORGANISATION ARBITRATION RULES (as from 1 October 2002) I. GENERAL PROVISIONS Abbreviated Expressions Article 1 In these Rules: Arbitration Agreement means
And, whereas, the objections and the suggestions received from the public on the said draft rules have been considered by the Central Government;
 *TO BE PUBLISHED IN THE GAZETTE OF INDIA, EXTRAORDINARY, PART II, SECTION 3, SUB SECTION (i)+ Government of India MINISTRY OF COMMERCE AND INDUSTRY (Department of Industrial Policy and Promotion) Notification
*TO BE PUBLISHED IN THE GAZETTE OF INDIA, EXTRAORDINARY, PART II, SECTION 3, SUB SECTION (i)+ Government of India MINISTRY OF COMMERCE AND INDUSTRY (Department of Industrial Policy and Promotion) Notification
INDIAN PATENT OFFICE
 Page 1 IN INDIAN PATENT OFFICE AS DESIGNATED (OR ELECTED) OFFICE CONTENTS THE ENTRY INTO THE NATIONAL PHASE SUMMARY THE PROCEDURE IN THE NATIONAL PHASE ANNEXES Fees... Annex IN.I Application for Grant
Page 1 IN INDIAN PATENT OFFICE AS DESIGNATED (OR ELECTED) OFFICE CONTENTS THE ENTRY INTO THE NATIONAL PHASE SUMMARY THE PROCEDURE IN THE NATIONAL PHASE ANNEXES Fees... Annex IN.I Application for Grant
Patent Term Extensions in Taiwan
 This article was published in the Markgraf Ergänzende Schutzzertifikate - Patent Term Extensions on 2015. Patent Term Extensions in Taiwan I. Introduction Ruth Fang, Lee and Li Attorneys at Law The patent
This article was published in the Markgraf Ergänzende Schutzzertifikate - Patent Term Extensions on 2015. Patent Term Extensions in Taiwan I. Introduction Ruth Fang, Lee and Li Attorneys at Law The patent
PRE-GRANT OPPOSITION POST-GRANT OPPOSITION
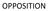 OPPOSITION TYPES OF OPPOSITION PRE-GRANT OPPOSITION [SEC 25(1)] POST-GRANT OPPOSITION [SEC. 25 (2)] REVOCATION[SECs 64 TO 66] GROUNDS FOR OPPOSITION UNDER SECTIONS 25(1) & 25 (2) That the applicant for
OPPOSITION TYPES OF OPPOSITION PRE-GRANT OPPOSITION [SEC 25(1)] POST-GRANT OPPOSITION [SEC. 25 (2)] REVOCATION[SECs 64 TO 66] GROUNDS FOR OPPOSITION UNDER SECTIONS 25(1) & 25 (2) That the applicant for
Considerations for the United States
 Considerations for the United States Speaker: Donald G. Lewis US Patent Attorney California Law Firm Leahy-Smith America Invents Act First Inventor to file, with grace period Derivation Actions Prior user
Considerations for the United States Speaker: Donald G. Lewis US Patent Attorney California Law Firm Leahy-Smith America Invents Act First Inventor to file, with grace period Derivation Actions Prior user
EGYPTIAN PATENT OFFICE
 PCT Applicant s Guide National Phase National Chapter EG Page 1 EGYPTIAN PATENT OFFICE AS DESIGNATED (OR ELECTED) OFFICE CONTENTS THE ENTRY INTO THE NATIONAL PHASE SUMMARY THE PROCEDURE IN THE NATIONAL
PCT Applicant s Guide National Phase National Chapter EG Page 1 EGYPTIAN PATENT OFFICE AS DESIGNATED (OR ELECTED) OFFICE CONTENTS THE ENTRY INTO THE NATIONAL PHASE SUMMARY THE PROCEDURE IN THE NATIONAL
Review of Current Status of Post-Grant Opposition System in Comparison with Invalidation Trial System
 Seiwa Patent & Law (IP Information Section) Dated April 29, 2016 Review of Current Status of Post-Grant Opposition System in Comparison with Invalidation Trial System Miyako Saito (patent attorney) and
Seiwa Patent & Law (IP Information Section) Dated April 29, 2016 Review of Current Status of Post-Grant Opposition System in Comparison with Invalidation Trial System Miyako Saito (patent attorney) and
The Patent Failure of Novartis with Gleevec
 1 The Patent Failure of Novartis with Gleevec The Indian Supreme Court s verdict on the Novartis patent application has garnered a lot of attention as having set a stringent standard of nonobviousness
1 The Patent Failure of Novartis with Gleevec The Indian Supreme Court s verdict on the Novartis patent application has garnered a lot of attention as having set a stringent standard of nonobviousness
LALL & SETHI ADVOCATES
 PATENT IN I. Filing of Application An application for patent can be made by completing and submitting a set of forms along with the prescribed fees with the Indian Patent Office (IPO). To file an application
PATENT IN I. Filing of Application An application for patent can be made by completing and submitting a set of forms along with the prescribed fees with the Indian Patent Office (IPO). To file an application
November Contents. Article Willful or deliberate suppression standard under Section 8 of the Patents Act. Ratio Decidendi News Nuggets
 An e-newsletter from Lakshmikumaran & Sridharan, New Delhi, India November 2014 / Issue 40 Contents Article Willful or deliberate suppression standard under Section 8 of the Patents Act Ratio Decidendi
An e-newsletter from Lakshmikumaran & Sridharan, New Delhi, India November 2014 / Issue 40 Contents Article Willful or deliberate suppression standard under Section 8 of the Patents Act Ratio Decidendi
IN THE SUPREME COURT OF INDIA CIVIL APPELLATE JURISDICTION CIVIL APPEAL NO(S) OF 2017 SPECIAL LEAVE PETITION (C) NO(S) OF 2016] Versus
![IN THE SUPREME COURT OF INDIA CIVIL APPELLATE JURISDICTION CIVIL APPEAL NO(S) OF 2017 SPECIAL LEAVE PETITION (C) NO(S) OF 2016] Versus IN THE SUPREME COURT OF INDIA CIVIL APPELLATE JURISDICTION CIVIL APPEAL NO(S) OF 2017 SPECIAL LEAVE PETITION (C) NO(S) OF 2016] Versus](/thumbs/90/102025175.jpg) 1 IN THE SUPREME COURT OF INDIA CIVIL APPELLATE JURISDICTION REPORTABLE CIVIL APPEAL NO(S). 9836 OF 2017 [@ SPECIAL LEAVE PETITION (C) NO(S). 34628 OF 2016] Haffkine Bio-Pharmaceutical Corporation Ltd.,
1 IN THE SUPREME COURT OF INDIA CIVIL APPELLATE JURISDICTION REPORTABLE CIVIL APPEAL NO(S). 9836 OF 2017 [@ SPECIAL LEAVE PETITION (C) NO(S). 34628 OF 2016] Haffkine Bio-Pharmaceutical Corporation Ltd.,
No.44. Special Issue. First Instance Judgment on the Patent Infringement Dispute Between IWNCOMM and Sony China. I. Summary of the Case
 No.44 Special Issue Case Express First instance judgment on the patent infringement dispute between IWNCOMM and Sony China First Instance Judgment on the Patent Infringement Dispute Between IWNCOMM and
No.44 Special Issue Case Express First instance judgment on the patent infringement dispute between IWNCOMM and Sony China First Instance Judgment on the Patent Infringement Dispute Between IWNCOMM and
T H E W O R L D J O U R N A L O N J U R I S T I C P O L I T Y. BOLAR EXEMPTION VS. DATA EXCLUSIVITY: RIGHT TO HEALTH vs RIGHT OF PATENT HOLDER
 BOLAR EXEMPTION VS. DATA EXCLUSIVITY: RIGHT TO HEALTH vs RIGHT OF PATENT HOLDER Rhea Roy Mammen M.S. Ramaiah College of Law, Bangalore Introduction Pharmaceutical Patent has seen an increasing conflict
BOLAR EXEMPTION VS. DATA EXCLUSIVITY: RIGHT TO HEALTH vs RIGHT OF PATENT HOLDER Rhea Roy Mammen M.S. Ramaiah College of Law, Bangalore Introduction Pharmaceutical Patent has seen an increasing conflict
2011 Foley & Lardner LLP Attorney Advertising Prior results do not guarantee a similar outcome Models used are not clients but may be representative
 2011 Foley & Lardner LLP Attorney Advertising Prior results do not guarantee a similar outcome Models used are not clients but may be representative of clients 321 N. Clark Street, Suite 2800, Chicago,
2011 Foley & Lardner LLP Attorney Advertising Prior results do not guarantee a similar outcome Models used are not clients but may be representative of clients 321 N. Clark Street, Suite 2800, Chicago,
Notification PART I CHAPTER I PRELIMINARY
 [TO BE PUBLISHED IN THE GAZZETTE OF INDIA, EXTRAORDINARY, PART II, SECTION 3, SUB-SECTION (i)] GOVERNMENT OF INDIA MINISTRY OF COMMERCE AND INDUSTRY (DEPARTMENT OF INDUSTRIAL POLICY AND PROMOTION) Notification
[TO BE PUBLISHED IN THE GAZZETTE OF INDIA, EXTRAORDINARY, PART II, SECTION 3, SUB-SECTION (i)] GOVERNMENT OF INDIA MINISTRY OF COMMERCE AND INDUSTRY (DEPARTMENT OF INDUSTRIAL POLICY AND PROMOTION) Notification
From The Editor s Desk
 From The Editor s Desk Dear Reader, Welcome to the fourth edition of the "IP India News"! We have entered a new year - here is wishing you a very happy new year and happy reading too. Shortly after entering
From The Editor s Desk Dear Reader, Welcome to the fourth edition of the "IP India News"! We have entered a new year - here is wishing you a very happy new year and happy reading too. Shortly after entering
DRAFT PATENT LAW OF GEORGIA CHAPTER I. GENERAL PROVISIONS
 DRAFT PATENT LAW OF GEORGIA CHAPTER I. GENERAL PROVISIONS ARTICLE 1 This Law regulates property and personal non-property relations formed in connection with the creation, legal protection and usage of
DRAFT PATENT LAW OF GEORGIA CHAPTER I. GENERAL PROVISIONS ARTICLE 1 This Law regulates property and personal non-property relations formed in connection with the creation, legal protection and usage of
Overview of Trial for Invalidation and Opposition Systems in Japan. March 2017 Trial and Appeal Department Japan Patent Office
 Overview of Trial for Invalidation and Opposition Systems in Japan March 2017 Trial and Appeal Department Japan Patent Office 1 Roles of Trial and Appeal Department of JPO Reviewing the examination ->
Overview of Trial for Invalidation and Opposition Systems in Japan March 2017 Trial and Appeal Department Japan Patent Office 1 Roles of Trial and Appeal Department of JPO Reviewing the examination ->
Patent Prosecution in View of The America Invents Act. Overview
 Patent Prosecution in View of The America Invents Act Courtenay C. Brinckerhoff David Dutcher Paul S. Hunter 2 Overview First-To-File (new 35 U.S.C. 102) Derivation Proceedings New Proceedings For Patent
Patent Prosecution in View of The America Invents Act Courtenay C. Brinckerhoff David Dutcher Paul S. Hunter 2 Overview First-To-File (new 35 U.S.C. 102) Derivation Proceedings New Proceedings For Patent
Law in the Global Marketplace: Intellectual Property and Related Issues FRAND in Europe: Huawei vs ZTE decision
 Law in the Global Marketplace: Intellectual Property and Related Issues FRAND in Europe: Huawei vs ZTE decision Hosted by: Overview Why the decision is important What does the Huawei vs ZTE decision say?
Law in the Global Marketplace: Intellectual Property and Related Issues FRAND in Europe: Huawei vs ZTE decision Hosted by: Overview Why the decision is important What does the Huawei vs ZTE decision say?
Notes and Disclaimers:
 THE PATENTS RULES, 2003 Notes and Disclaimers: 1. The e-version incorporates all the amendments in the Patents Act, 1970 and the Patent Rules, 2003 and is updated till 23-06-2017. 2. For details of every
THE PATENTS RULES, 2003 Notes and Disclaimers: 1. The e-version incorporates all the amendments in the Patents Act, 1970 and the Patent Rules, 2003 and is updated till 23-06-2017. 2. For details of every
ASIAN PATENT ATTORNEYS ASSOCIATION. 54th Council Meeting, Adelaide, Australia 2007 REPORT OF THE RECOGNIZED GROUP OF INDIA
 ASIAN PATENT ATTORNEYS ASSOCIATION 54th Council Meeting, Adelaide, Australia 2007 REPORT OF THE RECOGNIZED GROUP OF INDIA PRATHIBA M. SINGH Secretary, APAA (Indian Group) SINGH & SINGH ADVOCATES F-12,
ASIAN PATENT ATTORNEYS ASSOCIATION 54th Council Meeting, Adelaide, Australia 2007 REPORT OF THE RECOGNIZED GROUP OF INDIA PRATHIBA M. SINGH Secretary, APAA (Indian Group) SINGH & SINGH ADVOCATES F-12,
AUSTRIA Utility Model Law
 AUSTRIA Utility Model Law BGBl. No. 211/1994 as amended by BGBl. Nos. 175/1998, 143/2001, I 2004/149, I 2005/42, I 2005/130, I 2005/151, I 2007/81 and I 2009/126 TABLE OF CONTENTS I. GENERAL PROVISIONS
AUSTRIA Utility Model Law BGBl. No. 211/1994 as amended by BGBl. Nos. 175/1998, 143/2001, I 2004/149, I 2005/42, I 2005/130, I 2005/151, I 2007/81 and I 2009/126 TABLE OF CONTENTS I. GENERAL PROVISIONS
*IN THE HIGH COURT OF DELHI AT NEW DELHI. % Date of decision: 16 th February, Versus
 *IN THE HIGH COURT OF DELHI AT NEW DELHI + CM(M) No.815/2007 % Date of decision: 16 th February, 2010 OIL AND NATURAL GAS CORPORATION LTD.... Petitioner Through: Mr. V.N. Kaura with Ms. Paramjit Benipal
*IN THE HIGH COURT OF DELHI AT NEW DELHI + CM(M) No.815/2007 % Date of decision: 16 th February, 2010 OIL AND NATURAL GAS CORPORATION LTD.... Petitioner Through: Mr. V.N. Kaura with Ms. Paramjit Benipal
Construction of second medical use claims. The Hon. Mr Justice Richard Arnold
 Construction of second medical use claims The Hon. Mr Justice Richard Arnold The problem Claim 1 of European Patent (UK) No. 0 934 061 reads: Use of [pregabalin] or a pharmaceutically acceptable salt thereof
Construction of second medical use claims The Hon. Mr Justice Richard Arnold The problem Claim 1 of European Patent (UK) No. 0 934 061 reads: Use of [pregabalin] or a pharmaceutically acceptable salt thereof
CHAPTER 9 INVESTMENT. Section A
 CHAPTER 9 INVESTMENT Section A Article 9.1: Definitions For the purposes of this Chapter: Centre means the International Centre for Settlement of Investment Disputes (ICSID) established by the ICSID Convention;
CHAPTER 9 INVESTMENT Section A Article 9.1: Definitions For the purposes of this Chapter: Centre means the International Centre for Settlement of Investment Disputes (ICSID) established by the ICSID Convention;
SINGAPORE INTERNATIONAL ARBITRATION CENTRE (SIAC)
 GUIDE TO INTERNATIONAL ARBITRATION IN SINGAPORE INTERNATIONAL ARBITRATION CENTRE (SIAC) Written By S. Ravi Shankar Advocate on Record - Supreme Court of India National President of Arbitration Bar of India
GUIDE TO INTERNATIONAL ARBITRATION IN SINGAPORE INTERNATIONAL ARBITRATION CENTRE (SIAC) Written By S. Ravi Shankar Advocate on Record - Supreme Court of India National President of Arbitration Bar of India
FINAL REPORT THE PATENTS AND DESIGNS ACT, INTRODUCTION PATENTS
 FINAL REPORT ON THE PATENTS AND DESIGNS ACT, 200----- INTRODUCTION PATENTS In England grants of monopoly rights to exploit an invention by the inventor date back to the Elizabethan (Queen Elizabeth I)
FINAL REPORT ON THE PATENTS AND DESIGNS ACT, 200----- INTRODUCTION PATENTS In England grants of monopoly rights to exploit an invention by the inventor date back to the Elizabethan (Queen Elizabeth I)
Utility Model Law I. GENERAL PROVISIONS
 Utility Model Law Federal Law Gazette 1994/211 as amended by Federal Law Gazette I 1998/175, I 2001/143, I 2004/149, I 2005/42, I 2005/130, I 2005/151, I 2007/81 and I 2009/126 I. GENERAL PROVISIONS Subject
Utility Model Law Federal Law Gazette 1994/211 as amended by Federal Law Gazette I 1998/175, I 2001/143, I 2004/149, I 2005/42, I 2005/130, I 2005/151, I 2007/81 and I 2009/126 I. GENERAL PROVISIONS Subject
English Language Translation Entry into New Zealand PCT National Phase
 2009 Business Updates Request for postponement of acceptance under section 20(1) of the Patents Act 1953 Applicants may at any time prior to acceptance request that a patent application not be accepted
2009 Business Updates Request for postponement of acceptance under section 20(1) of the Patents Act 1953 Applicants may at any time prior to acceptance request that a patent application not be accepted
IN THE HIGH COURT OF DELHI AT NEW DELHI. % Judgment delivered on: versus M/S R.S. SALES CORPORATION & ANR
 IN THE HIGH COURT OF DELHI AT NEW DELHI % Judgment delivered on: 28.07.2016 + CS(COMM) 644/2016 ADITYA BIRLA NUVO LIMITED versus M/S R.S. SALES CORPORATION & ANR... Plaintiff... Defendants Advocates who
IN THE HIGH COURT OF DELHI AT NEW DELHI % Judgment delivered on: 28.07.2016 + CS(COMM) 644/2016 ADITYA BIRLA NUVO LIMITED versus M/S R.S. SALES CORPORATION & ANR... Plaintiff... Defendants Advocates who
Huawei v ZTE No More Need To Look At The Orange Book In SEP Disputes
 1 Huawei v ZTE No More Need To Look At The Orange Book In SEP Disputes By James Killick & Stratigoula Sakellariou 1 (White & Case) September 2015 Industry standards are crucial for economic development
1 Huawei v ZTE No More Need To Look At The Orange Book In SEP Disputes By James Killick & Stratigoula Sakellariou 1 (White & Case) September 2015 Industry standards are crucial for economic development
THE MICRO, SMALL AND MEDIUM ENTERPRISES DEVELOPMENT ACT, 2006 No. 27 of 2006
 THE MICRO, SMALL AND MEDIUM ENTERPRISES DEVELOPMENT ACT, 2006 No. 27 of 2006 [16th June, 2006.] An Act to provide for facilitating the promotion and development and enhancing the competitiveness of micro,
THE MICRO, SMALL AND MEDIUM ENTERPRISES DEVELOPMENT ACT, 2006 No. 27 of 2006 [16th June, 2006.] An Act to provide for facilitating the promotion and development and enhancing the competitiveness of micro,
Are Your Chinese Patents At Risk?
 October 2004 Are Your Chinese Patents At Risk? Viagra, the anti-impotence drug made by Pfizer, generated about $1.7 billion in worldwide sales last year. Viagra s active ingredient is a substance called
October 2004 Are Your Chinese Patents At Risk? Viagra, the anti-impotence drug made by Pfizer, generated about $1.7 billion in worldwide sales last year. Viagra s active ingredient is a substance called
IN THE HIGH COURT OF DELHI AT NEW DELHI SUBJECT : APPOINTMENT MATTER Date of decision: 11th July, 2012 W.P.(C) No.1343/1998.
 IN THE HIGH COURT OF DELHI AT NEW DELHI SUBJECT : APPOINTMENT MATTER Date of decision: 11th July, 2012 W.P.(C) No.1343/1998 SRI GURU TEGH BAHADUR KHALSA POST GRADUATE EVENING COLLEGE Through: None....
IN THE HIGH COURT OF DELHI AT NEW DELHI SUBJECT : APPOINTMENT MATTER Date of decision: 11th July, 2012 W.P.(C) No.1343/1998 SRI GURU TEGH BAHADUR KHALSA POST GRADUATE EVENING COLLEGE Through: None....
Case number 2011 (Wa) 38969
 Date February 28, 2013 Court Tokyo District Court, Case number 2011 (Wa) 38969 46th Civil Division A case in which the court found that an act of exercising the right to demand damages based on a patent
Date February 28, 2013 Court Tokyo District Court, Case number 2011 (Wa) 38969 46th Civil Division A case in which the court found that an act of exercising the right to demand damages based on a patent
The European patent system
 The European patent system Presenter: Dominique Winne Examiner (ICT) 7 November 2017 Contents EPC PCT Granting procedure at the 2 1 Optional The patent system yesterday and today Senate of Venice, 1474
The European patent system Presenter: Dominique Winne Examiner (ICT) 7 November 2017 Contents EPC PCT Granting procedure at the 2 1 Optional The patent system yesterday and today Senate of Venice, 1474
Damages and Remedies in Civil IP Cases An U.S. Perspective
 Damages and Remedies in Civil IP Cases An U.S. Perspective Elaine B. Gin Attorney - Advisor Office of Intellectual Property Policy and Enforcement US Patent & Trademark Office Every right has a remedy
Damages and Remedies in Civil IP Cases An U.S. Perspective Elaine B. Gin Attorney - Advisor Office of Intellectual Property Policy and Enforcement US Patent & Trademark Office Every right has a remedy
IN THE SUPREME COURT OF INDIA FERANI HOTELS PVT. LTD..APPELLANT. versus THE STATE INFORMATION COMMISSIONER GREATER MUMBAI & ORS..
 Reportable IN THE SUPREME COURT OF INDIA CIVIL APPELLATE JURISDICTION CIVIL APPEAL Nos.9064-9065 of 2018 [Arising out of SLP(C) Nos.32073-32074/2015] FERANI HOTELS PVT. LTD..APPELLANT versus THE STATE
Reportable IN THE SUPREME COURT OF INDIA CIVIL APPELLATE JURISDICTION CIVIL APPEAL Nos.9064-9065 of 2018 [Arising out of SLP(C) Nos.32073-32074/2015] FERANI HOTELS PVT. LTD..APPELLANT versus THE STATE
